It started in 1992, on the request of one of my students, who could not afford the zinc plate I asked them to purchase when we began the etching section of the printmaking class. Since ferric or stannous chloride was not acceptable to the director of the department, I tried using copper sulfate as I had found from past experience in trying to  copper plate the images on lithographic aluminium plates. The original formula from the Lithographic Technical Foundation called for the use of cupric chloride, which I could not get even from chemical stores at the university because it was an expensive material. Hoping that adding hydrochloric acid to copper sulfate I might get the necessary results, I found to my horror that the image was actually etched out instead of plated. Recalling this disaster, I went on to develop a mordant by adding hydrochloric acid to a solution of copper sulfate. We purchased large sheets of common aluminium and had them sheared to smaller size to sell to the students at cost to finish the class that year.
copper plate the images on lithographic aluminium plates. The original formula from the Lithographic Technical Foundation called for the use of cupric chloride, which I could not get even from chemical stores at the university because it was an expensive material. Hoping that adding hydrochloric acid to copper sulfate I might get the necessary results, I found to my horror that the image was actually etched out instead of plated. Recalling this disaster, I went on to develop a mordant by adding hydrochloric acid to a solution of copper sulfate. We purchased large sheets of common aluminium and had them sheared to smaller size to sell to the students at cost to finish the class that year.
 During the summer session class, I decided to remove the liquid acid and added salt to get the required chlorine molecules, ending up with cupreous chloride and sodium sulfate in solution. Because there now was little actual acid in the bath, particles of aluminium hydroxide formed that made the solution thick and cloudy, even though etching of the plate could proceed slowly. What was needed was an acid that would not attack aluminium but keep the pH below 7 to prevent formation of aluminium hydroxide. Again I looked for a dry material that could work and chose sodium bisulfate that forms weaker sulfurous acid, which I have used as a pickling solution in goldsmithing.
During the summer session class, I decided to remove the liquid acid and added salt to get the required chlorine molecules, ending up with cupreous chloride and sodium sulfate in solution. Because there now was little actual acid in the bath, particles of aluminium hydroxide formed that made the solution thick and cloudy, even though etching of the plate could proceed slowly. What was needed was an acid that would not attack aluminium but keep the pH below 7 to prevent formation of aluminium hydroxide. Again I looked for a dry material that could work and chose sodium bisulfate that forms weaker sulfurous acid, which I have used as a pickling solution in goldsmithing.
To fully understand what was going on, I approached the chemistry department, who became interested in the process as they realized it was much safer than the use of nitric acid, which had been used in the print department since its inception in the 1970’s. The professors assigned their students to work out the chemical reactions, which they gave me so that Dr. Bader and I submitted an article to LEONARDO, the refereed journal for the arts, in 1994. The article was accepted - but then the editors became concerned that copper compounds would be put into the environment and said they could not publish the piece. I convinced them that since normal etching of copper was done with nitric  acid and most likely much copper ended down the drain; but our process could eliminate that. Another group of referees were asked to review the article that was finally published in 1998. Since 1992, my process was available to printmakers through mail or my university website, so I had to answer many questions by e-mail from interested etchers. Etching done by Yoko Imabayashi.
acid and most likely much copper ended down the drain; but our process could eliminate that. Another group of referees were asked to review the article that was finally published in 1998. Since 1992, my process was available to printmakers through mail or my university website, so I had to answer many questions by e-mail from interested etchers. Etching done by Yoko Imabayashi.
Because of my interest in electronics, while surfing the Internet in 2007, I found a blog site in Australia in which hobbyist electronic workers started to use cupric chloride to etch their circuit boards instead of ferric chloride. It was a very detail account of how to prepare the starter solution from scrap copper wire, hydrochloric acid and hydrogen peroxide. The strength of the bath would increase with every board they etched and so was an efficient process for them. They did this because of the cost of cupric chloride and its’ unavailability in the community. The fact their bath regenerated with the availability of oxygen, made this the cheapest and safest method of etching copper.
electronic workers started to use cupric chloride to etch their circuit boards instead of ferric chloride. It was a very detail account of how to prepare the starter solution from scrap copper wire, hydrochloric acid and hydrogen peroxide. The strength of the bath would increase with every board they etched and so was an efficient process for them. They did this because of the cost of cupric chloride and its’ unavailability in the community. The fact their bath regenerated with the availability of oxygen, made this the cheapest and safest method of etching copper.
Realizing that 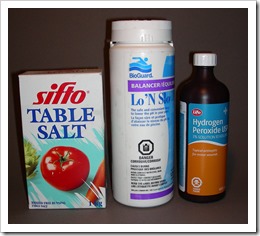 a strong enough hydrochloric acid can be made from salt and sodium bisulfate, I proceeded to do research into modifying my original copper sulfate etch to work on copper plates. By 2008 I had worked out the problems and contacted Alfons Bytautas at Edinburgh Printstudio; as I had been invited to do workshops in Ireland that year and hoped to visit him as well. Alfons had been working with my original copper sulfate mordant and now started to experiment with the cupric chloride solution and how well it can be regenerated. I published my work in Printmaking Today, Vol. 18, #2. Alfons followed up with his article in the next issue. What is most amazing in using these copper compounds to etch the metal, is the regeneration of the bath so that it is much cheaper to use and better for the environment. This seems to have been overlooked by those who have decided to etch zinc or aluminium plates – at a higher cost than need be to their budget.
a strong enough hydrochloric acid can be made from salt and sodium bisulfate, I proceeded to do research into modifying my original copper sulfate etch to work on copper plates. By 2008 I had worked out the problems and contacted Alfons Bytautas at Edinburgh Printstudio; as I had been invited to do workshops in Ireland that year and hoped to visit him as well. Alfons had been working with my original copper sulfate mordant and now started to experiment with the cupric chloride solution and how well it can be regenerated. I published my work in Printmaking Today, Vol. 18, #2. Alfons followed up with his article in the next issue. What is most amazing in using these copper compounds to etch the metal, is the regeneration of the bath so that it is much cheaper to use and better for the environment. This seems to have been overlooked by those who have decided to etch zinc or aluminium plates – at a higher cost than need be to their budget.
Over the last year, I have been exchanging e-mail with Fabiola Mercandetti in Rome, Italy. She and Marco had chosen to use the burin instead of etching plates with dangerous nitric acid - but they had found my websites and started to experiment with safer etching. Fabiola has been doing brilliant research into the historic use of copper compounds in etching and to my surprise; it goes back to before Goya’s use on his zinc etchings. All this time there has been this safer method for etching plates but the printmakers simply overlooked it and chose to stay with acids. She and several others realize that my publication on my websites and LEONARDO has reintroduced the process to this generation and it has been taken up by a few studios and teaching institutions because of their awareness of dangerous materials in our studio. It is another example of reinventing the wheel!
The reaction between copper and other metal depen ds on the electrochemical reduction/reaction potential of voltage between the metals concerned. With copper at +0.34 volts and aluminum at -1.66 volts, there is a potential difference of 2 volts, thus aluminium should etch faster than zinc with a difference of only 1.1 volt. With the presence of weak hydrochloric acid, it reacts with both of these metals to produce hydrogen gas in very a small amount to lift the resulting copper particles out of the etched line to float on the surface as long as it is not disturbed. There is no need for feathering, which actually breaks the bubbles and allows the copper particles to sink.
ds on the electrochemical reduction/reaction potential of voltage between the metals concerned. With copper at +0.34 volts and aluminum at -1.66 volts, there is a potential difference of 2 volts, thus aluminium should etch faster than zinc with a difference of only 1.1 volt. With the presence of weak hydrochloric acid, it reacts with both of these metals to produce hydrogen gas in very a small amount to lift the resulting copper particles out of the etched line to float on the surface as long as it is not disturbed. There is no need for feathering, which actually breaks the bubbles and allows the copper particles to sink.
While ma ny combinations of chemicals work, I found that one kilogram of copper sulfate added to 10-20 liters of water will make a good mordant, depending on how strong you want your bath. To this add salt until the solution turns green, indicating the presence of cuprous chloride. I have since found out that it is best to use pickling salt rather than common table salt that contains calcium silicate to prevent clumping in damp conditions. When table salt is used, insoluble calcium sulfate is formed, which is known as Plaster of Paris, used in casting projects. Technically pure salt can be purchased for softening water in the household and sodium bisulfate can be found at stores dealing with swimming pools and hot tubs. These compounds produce hydrochloric acid with a pH under 1.0.
ny combinations of chemicals work, I found that one kilogram of copper sulfate added to 10-20 liters of water will make a good mordant, depending on how strong you want your bath. To this add salt until the solution turns green, indicating the presence of cuprous chloride. I have since found out that it is best to use pickling salt rather than common table salt that contains calcium silicate to prevent clumping in damp conditions. When table salt is used, insoluble calcium sulfate is formed, which is known as Plaster of Paris, used in casting projects. Technically pure salt can be purchased for softening water in the household and sodium bisulfate can be found at stores dealing with swimming pools and hot tubs. These compounds produce hydrochloric acid with a pH under 1.0.
For etching both aluminium and zinc, I recommend using the original formula because of the hydrogen produced to keep the plate textures free for etching. Cupric chloride with copper plates produces no gas and the heavier dark compound sinks, making a vertical tank with bubbling air good for two reasons. One is to keep the plate surface free of the dark compound and the introduction of oxygen that helps regenerate the bath.
textures free for etching. Cupric chloride with copper plates produces no gas and the heavier dark compound sinks, making a vertical tank with bubbling air good for two reasons. One is to keep the plate surface free of the dark compound and the introduction of oxygen that helps regenerate the bath.
Since man y etchers prefer copper to the other metals, cupric chloride is the perfect answer for their studio. The commercial electronic industry has mostly turned to using cupric chloride over ferric chloride for a number of reasons. First, they don’t have to constantly purchase the ferric chloride and pay for its disposal; the regeneration of cupric chloride with air not only saves money, but with the increase of strength of the mordant, chemical companies are willing to buy the surplus produced on a daily basis. If it is respectable enough for these companies, it certainly should be looked at seriously by schools and large print shops dealing with etching.
y etchers prefer copper to the other metals, cupric chloride is the perfect answer for their studio. The commercial electronic industry has mostly turned to using cupric chloride over ferric chloride for a number of reasons. First, they don’t have to constantly purchase the ferric chloride and pay for its disposal; the regeneration of cupric chloride with air not only saves money, but with the increase of strength of the mordant, chemical companies are willing to buy the surplus produced on a daily basis. If it is respectable enough for these companies, it certainly should be looked at seriously by schools and large print shops dealing with etching.
Hydrogen peroxide comes at 3% in drugstores and 12% a t beauty salons. While the 3% works, the constant use of it for oxygen will dilute the solution too much, so a stronger version should be sought after. In health food stores, there is 35% available in Canada but may not be so in other countries because of it possible use in bomb making. I use the 35% that I keep in a refrigerator to help it keep its original strength, but never add the full strength to the bath as it releases too much oxygen that is just lost for reaction within the solution.
t beauty salons. While the 3% works, the constant use of it for oxygen will dilute the solution too much, so a stronger version should be sought after. In health food stores, there is 35% available in Canada but may not be so in other countries because of it possible use in bomb making. I use the 35% that I keep in a refrigerator to help it keep its original strength, but never add the full strength to the bath as it releases too much oxygen that is just lost for reaction within the solution.
If peroxide is not available, then oxygen containing compounds such as potassium or sodium chlorate can be used, but the constant addition produces too much of other sodium/potassium compounds that can impede the mordents’ effectiveness. From time to time hydrogen is required for the reaction, so the pH can to be lowered with hydrochloric acid, but I still recommend you use the safer dry materials of salt and sodium bisulfate.
Both a pH me ter and hydrometer would be helpful if you need to get into producing a constant strength mordant. Inexpensive pocket pH meter can be found on the Internet and hydrometers used for testing car batteries can be purchased at most automotive suppliers. Fabiola has found a US patent (#2,908,557) from 1959 that outlines a new method of etching copper, in which I found that concentrations of hydrogen, chlorine and oxygen molecules can range by a great deal and still etch the metal efficiently. As long as sufficient amounts are present, an excess seemed to not trouble the patentees.
ter and hydrometer would be helpful if you need to get into producing a constant strength mordant. Inexpensive pocket pH meter can be found on the Internet and hydrometers used for testing car batteries can be purchased at most automotive suppliers. Fabiola has found a US patent (#2,908,557) from 1959 that outlines a new method of etching copper, in which I found that concentrations of hydrogen, chlorine and oxygen molecules can range by a great deal and still etch the metal efficiently. As long as sufficient amounts are present, an excess seemed to not trouble the patentees.
Alfons has researched cupric chloride very thoroughly and has published his results: Salt -175 grams and sodium bisulfate -125 grams in one liter of water. Add 200 ml of hydrogen peroxide and 200 grams of copper sulfate. This produces a mordant with specific gravity of 1.2609 and pH of 1.4. My research mixtures had a specific gravity of 1.200 to 1.305 and pH anywhere between .98 and 2.0. With a lower pH, more hydrogen is formed so the aluminium and zinc plates also etch faster. When first mixed the color is a beautiful leaf green and gets darker as more cupric chloride is formed, eventually becoming a dark brown when it stops reacting with the copper. The addition of peroxide will quickly return the bright green color and the bath is regenerated.
Disposing of mordant
There might be a time when you want to stop etching and get rid of the bath safely down the drain. Since copper compounds are toxic to fish and not remove d from the discharged water, it is simple matter to remove all the copper by putting in a scrap piece of aluminium until all the particles are at the bottom and there is still much of the scrap metal left. The copper particles ca
d from the discharged water, it is simple matter to remove all the copper by putting in a scrap piece of aluminium until all the particles are at the bottom and there is still much of the scrap metal left. The copper particles ca n be filtered out with a common coffee filter and a proper funnel, leaving aluminium hydroxide in solution. The hydroxide can be removed by adding a base such as washing soda or borax. The white hydroxide can also be filtered out with a coffee filter or synthetic cloth as I have done. The remaining liquid has no toxic compounds and can go down the drain.
n be filtered out with a common coffee filter and a proper funnel, leaving aluminium hydroxide in solution. The hydroxide can be removed by adding a base such as washing soda or borax. The white hydroxide can also be filtered out with a coffee filter or synthetic cloth as I have done. The remaining liquid has no toxic compounds and can go down the drain.
If you were etching zinc, the zinc chloride produced is soluble in w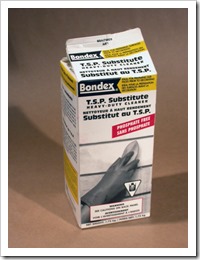 ater and should not be put down the drain. To remove the zinc, add sodium silicate or metasilicate to form zinc silicate that is whitish insoluble material that can be filtered out.
ater and should not be put down the drain. To remove the zinc, add sodium silicate or metasilicate to form zinc silicate that is whitish insoluble material that can be filtered out.
If copper plates were used, scrap aluminium will remove the copper like it does with cuprous chloride and the lesser amount of aluminum hydroxide formed can be filtered out as well.
Where to find materials
Much depends on the size of your community and number of major suppliers with stores. Here are suggestions for the least expensive products but good for the processes:
· Sodium bisulfate – Swimming pool and hot tub dealers, jewelry/craft stores. Dry crystals that produce milder sulfuric (sulfurous) acid in water.
· Sodium silicate- Pottery clay suppliers; used to modify clay. A viscous alkaline liquid that dries waterproof.
· Sodium metasilicate- House hold products in hardware and groceries. A dry powder soluble in water to replace phosphate powder that is bad for the environment. Look for ingredients on the box as some contain only washing soda. Bondex at Home Depot is my supply.
· Copper sulfate- Gardening supply or some hardware stores. Used to keep algae down in dugouts.
· Salt- Pickling salt at groceries or salt for use in household water softeners. Don’t use large pellets supplied by some softener companies. Salt must be free of calcium silicate.
· Hydrogen peroxide- Use at least 12% available as hair bleach in beauty salons. Health food stores might have 35% that you can dilute for adding to mordant.
· Chlorate compounds- Chemistry supplier but may be a bit more expensive and detrimental to the longtime effectiveness of the solution.
Interesting web sites with more information
Instead of going into all the fine detail of why and how copper compounds can etch metals more safely and just as effectively as acidic mordants , I recommend you go to the following websites where there is much information over many articles written as the processes were being developed.
· http://www.ndiprintmaking.ca/?paged=2— My personal website that blogs recent developments or items of interest in the arts.
· http://homepage.usask.ca/~nis715/ . This is my site at the university of Saskatchewan and has important information on all my innovations.
· http://members.optusnet.com.au/~eseychell/PCB/etching_CuCl/index.html. This is an excellent site to explain the chemistry of etching copper, starting with hydrochloric acid and hydrogen peroxide.
A Google search for “Copper etching with cupric chloride†will give you many commercial circuit board manufactures methods that might be of interest to you.
